A 36-year-old male patient was admitted to the emergency
department with abdominal and left side pain 1.5 hours after
an in-vehicle traffic accident. The patient had no history
of comorbidity or surgery. The patient did not receive any
anticoagulant or antiplatelet therapy prior to the trauma. Vital
signs of the patient were stable (Blood pressure 145/100
mmHg, pulse 98 beats/min, and temperature 37.2°C). Physical
examination revealed no additional pathology except left side
pain and left upper quadrant tenderness in deep palpation.
White blood cells were found to be 17.330/mm3, hemoglobin
level 17.34 mg/dL, and hematocrit 48.83% in the complete
blood count examination. There was no abnormality in the
biochemical analysis. An evaluation focused on sonography
for trauma (FAST) in the emergency department was negative.
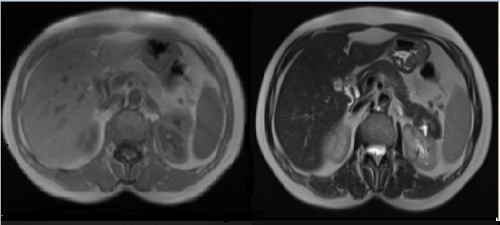
Contrast-enhanced thoracoabdominal computed tomography
(CT) scan was performed. Abdominal CT revealed a 44x42 mm left central hyperdense and peripheral hypodense adrenal mass
(63 hounsfield unit) and periadrenal fat strands. Additionally, a
cortical cyst was observed in the upper pole of the left kidney
(Figure 1). The lesion was evaluated as an adrenal hematoma.
No other injuries were detected, especially no injury to the
spleen or kidney. No rib or spine fractures were observed. The
patient was treated conservatively with bed rest, parenteral fluid,
antibiotherapy, and analgesics. Hemoglobin and biochemical
parameters remained constant. Endocrinology consultation was
requested for adrenal insufficiency. Endocrinological evaluations
revealed no pathology. The control abdominal CT performed 7
days later showed that the hematoma did not progress (40x37
mm, central density is prominent) (Figure 2). The patient was
discharged without any problems. An abdominal CT scan was
planned to evaluate the resolution of the adrenal hematoma 1
month later. The patient was asymptomatic during the follow-up. No abscess or infection format was observed. Informed
written consent was obtained from the patient for this report.
Figure 1: A- Left adrenal hematoma 44x42 mm attenuated central hyperdense and peripheral hypodense round mass in the adrenal gland, B- Periadrenal fat stranding,
C- Left kidney upper pole simple cortical cyst (Axial view of enhanced abdominal CT scan)
Figure 2: Control abdominal CT; regression of hematoma and increased appearance of
central hyperdensity (Axial view of enhanced abdominal CT scan)
Adrenal gland injury is a rare clinical picture caused by
motor vehicle accidents, sports injuries, or blunt abdominal
trauma after falling [–]. Isolated adrenal gland injury is rare
due to its small size, deep retroperitoneal position on the upper
abdomen, and presence of full-fat tissue around it. Most adrenal
gland injuries are associated with multiple adjacent skeletal and
organ injuries [].
Adrenal gland injuries have been reported in approximately
2-3% of all thoracoabdominal injuries []. Unilateral adrenal
injuries occur 5 times more on the right side than on the left
(77% versus 15%), and bilateral adrenal injuries occur in 8% of
cases in trauma [].
Being usually silent and self-limiting, it does not require
major operative intervention. However, it may be potentially
life-threatening in some cases. The most common symptom is
pain; other clinical presentations vary greatly, and it does not
produce any specific symptoms or biomarkers. Abdominal
pain, side pain, nausea, vomiting, hypotension, hypertension, a
palpable side mass, agitation, mental status changes, and lowgrade
fever may occur []. The emergency physician should be
aware of the possibility of organ damage associated with adrenal
injury and the potential for adrenal insufficiency especially if an
unusual complaint is presented after blunt trauma (unexplained
hypotension, electrolyte disorder, and pain that does not go away
despite analgesics) [].
Although ultrasonography is noninvasive, easily accessible,
and inexpensive, it is dependent on the person and can sometimes
be inadequate when evaluating retroperitoneal organs. CT
is the gold standard for detecting adrenal gland injury as in
all trauma cases []. CT scan findings of adrenal gland injury
include hyperdensity, periadrenal fat stranding infiltration, and
ipsilateral diaphragmatic crural thickening [,]. Furthermore,
the need to monitor and rule out an underlying adrenal neoplasm
should be taken into account in these patients due to possible
bleeding to a pre-existing adrenal mass [].
Surgery (adrenalectomy) and interventional radiologic
procedures (embolization) may be needed although most adrenal
gland injuries are treated conservatively. Treatment depends on
the hemodynamic condition of the patient, the severity of the
gland damage, bilateral gland involvement, and the extent of
bleeding within the gland [].
Ethics Committee Approval: N / A.
Informed Consent: An informed consent was obtained from
the patient.
Publication: The results of the study were not published in full
or in part in form of abstracts.
Peer-review: Externally peer-reviewed.
Conflict of Interest: The authors declare that they have no
conflict of interest.
Financial Disclosure: The authors declare that this study
received no financial support.